AbbVie Porter's Five Forces Analysis
Fully Editable
Tailor To Your Needs In Excel Or Sheets
Professional Design
Trusted, Industry-Standard Templates
Pre-Built
For Quick And Efficient Use
No Expertise Is Needed
Easy To Follow
AbbVie Bundle
What is included in the product
Pinpoints competitive pressures impacting AbbVie, including rivalry, supplier power, and potential threats.
Quickly identify threats and opportunities within the pharmaceutical market, improving decision-making.
Preview Before You Purchase
AbbVie Porter's Five Forces Analysis
This is the complete AbbVie Porter's Five Forces analysis you'll receive. The preview displays the full document, encompassing competitive rivalry, supplier power, buyer power, threat of substitution, and threat of new entrants.
Porter's Five Forces Analysis Template
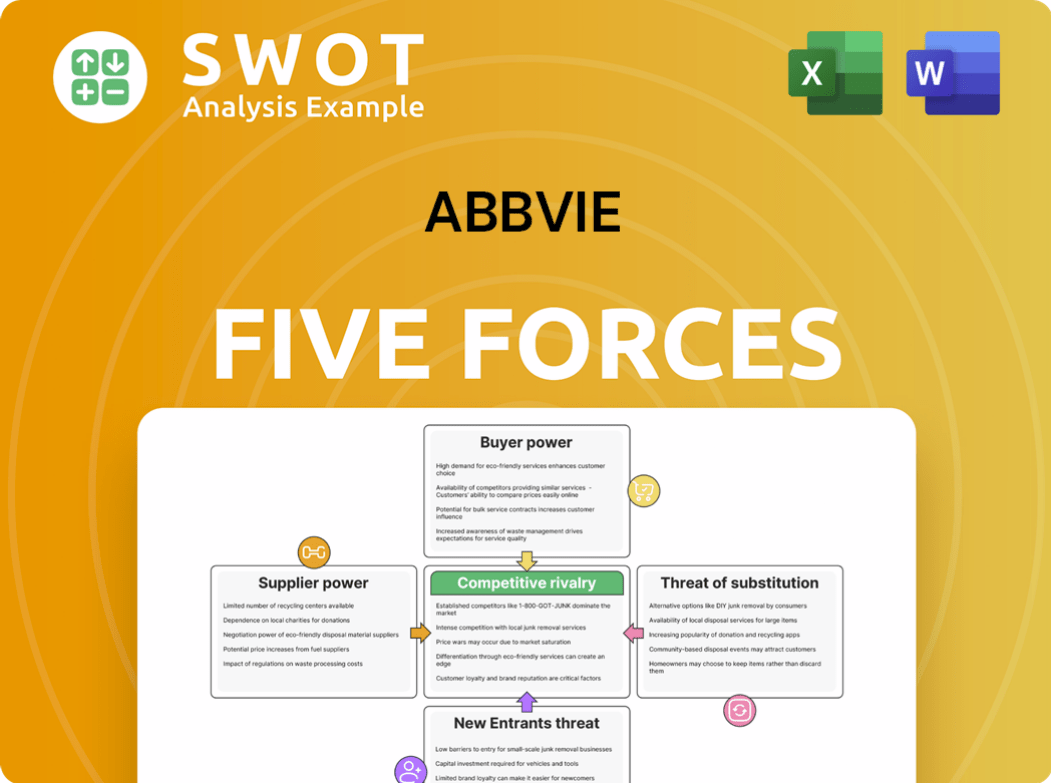
AbbVie faces intense competition, particularly from generic drug manufacturers and biosimilar rivals. Buyer power is moderate, influenced by insurance companies and healthcare providers negotiating prices. The threat of new entrants is relatively low due to high barriers to entry. Substitute products, such as other treatments for similar conditions, pose a moderate threat. Supplier power is manageable, as AbbVie has diverse suppliers.
Our full Porter's Five Forces report goes deeper—offering a data-driven framework to understand AbbVie's real business risks and market opportunities.
Suppliers Bargaining Power
AbbVie's reliance on specialized suppliers, especially for APIs, concentrates power. Limited suppliers, particularly for hard-to-source ingredients, increase their leverage. In 2023, AbbVie depended on 12-15 key global suppliers for essential raw materials. This concentration could impact production costs and flexibility.
High switching costs significantly bolster supplier bargaining power in the pharmaceutical sector. AbbVie faces substantial challenges when changing suppliers of pharmaceutical-grade materials. These challenges arise from rigorous regulatory demands. Switching costs can range from $2.5 million to $5.7 million per supplier qualification process.
Regulatory compliance introduces significant challenges for AbbVie, with approval processes potentially spanning 18-24 months. Compliance certification costs can range from $750,000 to $1.2 million. Quality validation expenses add another $450,000 to $850,000 to the switch costs. These factors strengthen supplier power by limiting AbbVie's options.
Geographic Concentration
AbbVie's reliance on suppliers is influenced by geographic concentration, particularly in Asia-Pacific and Europe. This concentration increases supplier bargaining power due to potential disruptions from geopolitical events or policy changes. In 2023, roughly 45-50% of AbbVie's suppliers were based in the Asia-Pacific region. This geographic setup makes AbbVie susceptible to supply chain vulnerabilities.
- Asia-Pacific suppliers: 45-50% in 2023.
- Geopolitical risk: Trade policies and events.
- Supply chain: Vulnerability due to concentration.
- Geographic factor: Key for supplier power.
Supplier Influence
AbbVie's suppliers wield moderate influence. Supplier concentration metrics show that the top three suppliers controlled about 40-45% of essential raw materials in 2023. This concentration enables suppliers to impact pricing and supply terms, affecting AbbVie's profitability.
- Supplier concentration: Top 3 suppliers control 40-45% of key raw materials.
- Pricing power: Suppliers can influence pricing due to their control.
- Profitability impact: Supplier actions can affect AbbVie's profit margins.
AbbVie faces supplier power due to its dependence on specialized, often geographically concentrated, suppliers. High switching costs, driven by regulatory demands and validation processes, further strengthen suppliers' leverage. In 2023, AbbVie sourced approximately 45-50% of its supplies from the Asia-Pacific region, making it vulnerable to disruptions.
| Factor | Impact | Data (2023) |
|---|---|---|
| Supplier Concentration | Increased bargaining power | Top 3 suppliers controlled 40-45% of key raw materials. |
| Geographic Concentration | Vulnerability to disruptions | 45-50% of suppliers based in Asia-Pacific. |
| Switching Costs | Limits AbbVie's options | Qualification costs: $2.5M - $5.7M per supplier. |
Customers Bargaining Power
Buyers of AbbVie's products, such as hospitals and insurance companies, have moderate bargaining power. This power is lessened for drugs treating critical conditions. In 2024, AbbVie’s blockbuster drug Humira faced biosimilar competition, impacting its pricing power. The introduction of biosimilars has increased buyer leverage, decreasing AbbVie's ability to set prices.
AbbVie benefits from limited substitutes for many drugs, especially in critical treatments. This gives the company leverage over buyers, as alternatives are scarce. This is especially true for complex medical conditions. However, the rise of generic drugs is starting to shift this dynamic. In 2024, generic competition impacted sales significantly.
AbbVie operates in a highly regulated pharmaceutical industry, where products must gain approval from health authorities like the FDA before being sold. This regulatory environment constrains customer choices, thereby diminishing their bargaining power. For example, in 2024, it took an average of 10-12 years and over $2 billion to bring a new drug to market. These regulations also create significant barriers to entry for potential substitutes, further limiting customer options and power.
Patent Protection
AbbVie's patent protection significantly influences its customer bargaining power. Patents shield drugs from generic competition, granting AbbVie pricing power due to limited alternatives. The expiration of patents, like Humira's, boosts buyer options with generics, lowering AbbVie's power. This dynamic impacts revenue streams and market share. In 2024, Humira's biosimilar competition affected sales.
- Patent protection limits customer choices.
- Patent expiration increases buyer options.
- Humira's patent expiry impacted sales.
- AbbVie's pricing power fluctuates with patents.
Generic Alternatives
The bargaining power of customers increases significantly when generic alternatives become available. After patents expire, generic versions of drugs like Humira enter the market, giving buyers more choices. This competition erodes AbbVie's pricing power, as seen with Humira's biosimilars. Pharmaceutical pricing depends on clinical outcomes, competition, and regulations.
- Humira's U.S. sales decreased by 32.3% in 2023 due to biosimilar competition.
- By 2024, multiple biosimilars for Humira are available in the U.S., intensifying price pressure.
- AbbVie's overall revenue in 2023 was $54.3 billion, reflecting the impact of generic competition.
- Price erosion is a key factor in contract negotiations.
Customers' bargaining power for AbbVie hinges on drug availability. Patent protection limits customer choices, giving AbbVie pricing power. However, patent expiration, like Humira's in 2023, boosts buyer options with generics, increasing their power.
| Metric | 2023 Data | Impact |
|---|---|---|
| Humira U.S. Sales Decline | 32.3% | Increased buyer leverage |
| AbbVie's Revenue (2023) | $54.3 billion | Reflects generic competition impact |
| Biosimilar Availability (2024) | Multiple in U.S. | Intensified price pressure |
Rivalry Among Competitors
The pharmaceutical industry is fiercely competitive. AbbVie competes with big and small biotech firms. This leads to intense rivalry, pressuring profitability. AbbVie's market share in Q4 2024 was 7.16%, highlighting the competitive environment.
The pharmaceutical industry is a battlefield, with companies fiercely vying for market share. AbbVie faces stiff competition in key areas like immunology and oncology, pushing for innovation. In 2024, AbbVie's top competitors include Johnson & Johnson and Pfizer. The company's success hinges on its ability to outmaneuver rivals through product development and marketing.
Patent expirations intensify competitive rivalry in the pharmaceutical industry. When patents on blockbuster drugs expire, such as AbbVie's Humira in 2023, generic and biosimilar manufacturers enter the market. This leads to increased competition and often price erosion. Humira's 2023 U.S. sales decreased by 32.2%, reflecting the impact of biosimilar competition.
R&D Investment
Competitive rivalry in the pharmaceutical industry is significantly driven by R&D investments, as companies strive for innovative products. High R&D spending indicates intense competition. AbbVie's 2023 R&D expenditure was $7.4 billion, or 17.2% of revenue, showcasing its commitment. This competitive landscape is further highlighted by similar investments from rivals like Pfizer and Merck.
- AbbVie's R&D spending in 2023: $7.4 billion
- AbbVie's R&D as % of revenue: 17.2%
- Pfizer's R&D spending in 2023: $10.8 billion
- Merck's R&D spending in 2023: $13.2 billion
Regulatory Pressures
AbbVie contends with regulatory scrutiny and pricing pressures, especially in the U.S. Healthcare policies and pricing practices affect drug pricing and market access, increasing competition among pharmaceutical companies. These pressures can significantly influence profitability and market strategies. For instance, the Inflation Reduction Act of 2022 is impacting drug pricing.
- The Inflation Reduction Act (IRA) allows Medicare to negotiate drug prices, starting with 10 drugs in 2026, potentially affecting AbbVie's revenue.
- AbbVie's Humira faced biosimilar competition, affecting its sales and market share.
- In 2023, AbbVie's net revenues were approximately $54.3 billion.
Competition in pharmaceuticals is intense, affecting AbbVie's market position. AbbVie's Q4 2024 market share was 7.16%, reflecting this rivalry. R&D spending, like AbbVie's $7.4B in 2023, fuels the competition. Patent expirations also intensify competitive pressures significantly.
| Metric | AbbVie (2023) | Pfizer (2023) |
|---|---|---|
| R&D Spend ($B) | 7.4 | 10.8 |
| R&D % of Revenue | 17.2% | N/A |
| Net Revenue ($B) | 54.3 | 58.5 |
SSubstitutes Threaten
AbbVie encounters a strong threat from emerging biologics and alternative treatments. The global biologics market is forecasted to hit $568.5 billion in 2024, highlighting the competition. Humira, AbbVie's key drug, faced biosimilar rivalry, diminishing its market share from 70% to 48% between 2019-2023.
The generic drug market poses a significant threat to AbbVie's branded pharmaceuticals. In 2024, this market is valued at $492.4 billion, growing at a 6.3% CAGR. Generic drugs in immunology have a 24.6% market share. They offer 80-85% price reductions compared to brand-name drugs. There are nine FDA-approved biosimilars for Humira.
Emerging therapeutic technologies pose a threat to AbbVie. The CRISPR gene editing market is forecast to hit $12.8 billion in 2024, impacting treatment methods. Gene therapy is valued at $23.4 billion, while RNA therapeutics are at $7.6 billion. These alternatives present a moderate to high potential impact on AbbVie's market position.
Personalized Medicine
Personalized medicine poses a growing threat to AbbVie's traditional pharmaceutical model. The market is substantial, projected to hit $796.8 billion in 2024. This shift towards tailored treatments could lessen the demand for broad-spectrum drugs. The high growth rate of 11.2% in this area, shows its rapid expansion.
- Market size: $796.8 billion by 2024.
- Growth rate: 11.2%.
- Genomic testing market: $62.3 billion.
- Precision medicine in oncology adoption: 42%.
Clinical Efficacy
The threat of substitutes for AbbVie's drugs hinges on their clinical efficacy relative to alternatives. Superior clinical outcomes reduce the likelihood of patients switching to substitutes. In 2024, AbbVie's Humira faced biosimilar competition, highlighting this dynamic. Strong efficacy data helps maintain market share and pricing power. This is crucial in competitive therapeutic areas.
- Humira's U.S. sales declined by 32.2% in 2023 due to biosimilar competition.
- Skyrizi and Rinvoq, AbbVie's newer drugs, are crucial for offsetting losses from Humira's decline.
- Clinical trials and real-world evidence demonstrating superior efficacy are vital to combat substitution threats.
AbbVie faces considerable threats from various substitutes. The personalized medicine market, valued at $796.8 billion in 2024, poses a significant challenge. Competition from biosimilars and generics, like Humira's decline, further intensifies this threat.
| Substitute Type | Market Size (2024) | Impact on AbbVie |
|---|---|---|
| Biosimilars/Generics | $492.4 billion | High; Erosion of market share and revenue |
| Personalized Medicine | $796.8 billion | Moderate; Shift in treatment approaches |
| Emerging Therapies (e.g., CRISPR) | Various (e.g., CRISPR $12.8B) | Moderate; Potential for alternative treatments |
Entrants Threaten
AbbVie benefits from high entry barriers, a significant advantage in the pharmaceutical industry. Developing a new drug costs around $2.6 billion. These massive costs make it tough for new firms to compete. This protects AbbVie's market position.
Developing new drugs demands massive investments in research, clinical trials, and regulatory approvals. High R&D costs create a significant barrier for new pharmaceutical companies. The process is slow and risky, with failure rates for drug candidates reaching approximately 90%. In 2024, the average cost to bring a new drug to market was estimated to be over $2.6 billion, according to the Tufts Center for the Study of Drug Development.
Navigating regulatory hurdles and obtaining approvals from health authorities pose a major challenge for new pharmaceutical entrants. Stringent requirements, including extensive clinical trials, create significant barriers. The approval process can take several years, adding to the complexity. For example, in 2024, the FDA approved approximately 50 new drugs, a testament to the rigorous process.
Supply Chain Complexity
The pharmaceutical industry's intricate global supply chains, logistics, and regulatory compliance pose significant barriers. New entrants struggle with these complexities, especially in establishing distribution networks. These established relationships are crucial for market access, hindering new companies. In 2024, the average time to bring a new drug to market was over a decade, reflecting these challenges.
- Global supply chains involve multiple stages.
- Regulatory hurdles slow market entry.
- Established distribution networks are key.
- High compliance costs are a barrier.
Brand Recognition
AbbVie, with its established presence, enjoys significant brand recognition, a key advantage against new entrants. Building trust with healthcare providers and patients requires considerable time and marketing investment, which is a hurdle for new companies. This brand loyalty translates to a competitive edge, making it challenging for newcomers to capture market share quickly.
- AbbVie's strong brand recognition provides a significant barrier to entry.
- New entrants face high costs and time to build brand trust.
- Customer loyalty to established brands limits market access for new companies.
- Marketing and promotion are major investments for new entrants.
AbbVie faces minimal threat from new entrants due to high barriers. The average cost to develop a new drug reached over $2.6 billion in 2024. Rigorous regulatory hurdles and complex supply chains further deter newcomers.
| Barrier | Impact |
|---|---|
| R&D Costs | ~$2.6B per drug (2024) |
| Regulatory | FDA approved ~50 drugs (2024) |
| Supply Chain | Over a decade to market (2024) |
Porter's Five Forces Analysis Data Sources
We leverage SEC filings, industry reports, and market analysis databases to inform our AbbVie assessment.